When given a problem to solve using stoich, you must always balance the chemical equation first If you don't know how, refer to our last chapter in this beautiful website. If you already have that skill, awesome
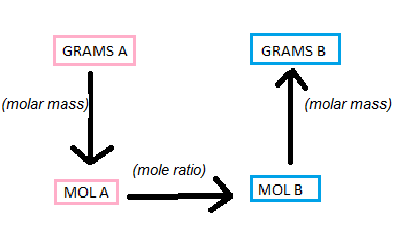
The Mole Box
The Mole Box
|
In your chemical equations you'll be working with two chemicals at a time, trying to figure out how to change one form of substance A to another form of substance B.
A universal unit for any substance in the world in the mole because it's the same for every element (6.02 X 10^-23), you must always convert your substance to moles before anything other step to that conversion is possible. |
ONE STEP: Grams A --> Moles A (vice versa) , Moles A --> Moles B (vice versa) , Moles B --> Grams B(vice versa)
TWO STEPS: Grams A --> Moles B (vice versa) , Moles A --> Grams B (vice versa)
THREE STEPS: Grams A --> Grams B (vice versa)
Gram to Mole/ Gram to Mole
As you can see in the picture, this process involves the use of molar mass. Molar mass is how many grams one mole of a substance contains. You get the molar mass by 1) Finding each respective element on the periodic table
2) Multipling it's atomic number on the table by it's coefficient in the equation
3) When dealing with compounds and/or molecules, add the sums together.
Ex) Molar Mass of H20?
Number of H: 1
Coefficient of H in molecule: 2
Molar mass of H in this equation: 1 X 2 = 2
Number of Oxygen: 16
Coefficient of O in this molecule:1
Molar mass of O in this equation: 16 x 1 = 16
Molar mass of molecule: 2 + 16 = 18g
The Molar mass of water is 18g!!
When converting from Grams of substance A to moles of substance A, simply divide the grams you start with by the substances respective molar mass and you'l have the number of moles! When going from mole to grams, simply multiply by the molar mass to go back to grams!
6g H20 = __ moles H20?? 6g H20 x 1 mol H20 = .33 moles of H20
18g H20
100 moles of H20 = __ grams H20??? 100mol H20 x 18g H20 = 1800 g of H20
1 mol H20
As you can see in the picture, this process involves the use of molar mass. Molar mass is how many grams one mole of a substance contains. You get the molar mass by 1) Finding each respective element on the periodic table
2) Multipling it's atomic number on the table by it's coefficient in the equation
3) When dealing with compounds and/or molecules, add the sums together.
Ex) Molar Mass of H20?
Number of H: 1
Coefficient of H in molecule: 2
Molar mass of H in this equation: 1 X 2 = 2
Number of Oxygen: 16
Coefficient of O in this molecule:1
Molar mass of O in this equation: 16 x 1 = 16
Molar mass of molecule: 2 + 16 = 18g
The Molar mass of water is 18g!!
When converting from Grams of substance A to moles of substance A, simply divide the grams you start with by the substances respective molar mass and you'l have the number of moles! When going from mole to grams, simply multiply by the molar mass to go back to grams!
6g H20 = __ moles H20?? 6g H20 x 1 mol H20 = .33 moles of H20
18g H20
100 moles of H20 = __ grams H20??? 100mol H20 x 18g H20 = 1800 g of H20
1 mol H20
Mole to Mole
Refers to the mole ratio you'll have set up with your balanced chemical equation
How many moles of __ is used with ___ moles of ___ ?
2H20 = 2H2 + 02
How many moles of Oxygen are present when 2 moles of H20 splits up?
2 mol H20 X 1 mol O2 = 1 mol 02
2 moles H20
Gram to Gram
These are the lengthiest questions you'll get asked. 3 step problems involve using the three different equations we just went over;
1) Gram A - Mol A
2) Mol A - Mol B
3) Mol B - Gram A
It's simply more dimensional analysis
Ex) 2 Na + Cl2 = 2 NaCl
How many grams of NaCl will be produced if you are given 80.00 g of Cl-1?
80.00g Cl x 1 mol Cl x 2 mol NaCl x (23g + 36g) = 59 g NaCl = 131.1 g NaCl
(36g + 36g) = 72 g Cl (MOLAR MASS) 1 mol Cl (MOLE RATIO) 1 mol NaCl (MOLAR MASS)
Refers to the mole ratio you'll have set up with your balanced chemical equation
How many moles of __ is used with ___ moles of ___ ?
2H20 = 2H2 + 02
How many moles of Oxygen are present when 2 moles of H20 splits up?
2 mol H20 X 1 mol O2 = 1 mol 02
2 moles H20
Gram to Gram
These are the lengthiest questions you'll get asked. 3 step problems involve using the three different equations we just went over;
1) Gram A - Mol A
2) Mol A - Mol B
3) Mol B - Gram A
It's simply more dimensional analysis
Ex) 2 Na + Cl2 = 2 NaCl
How many grams of NaCl will be produced if you are given 80.00 g of Cl-1?
80.00g Cl x 1 mol Cl x 2 mol NaCl x (23g + 36g) = 59 g NaCl = 131.1 g NaCl
(36g + 36g) = 72 g Cl (MOLAR MASS) 1 mol Cl (MOLE RATIO) 1 mol NaCl (MOLAR MASS)