|
In chemistry, the limiting reagent is the substance which is totally consumed once the chemical reaction has taken place. Since this substance is used up first, it limits the amount of product formed considering the reaction is unable to continue reacting without it. Therefore, the limiting reagent must be identified before you can calculate how much the reaction will yield.
____________________________________________________________ To better understand the concept, think of a recipe. Peanut Butter Cookie recipe: 1C. peanut butter + 1/2 C. sugar + 1 egg --> 1 batch At home I have 3 eggs, 4 cups of sugar and 2 cups of peanut butter. How many batches of cookies can I make? 2 c. PB X 1 batch 4 c. sugar X 1 batch ---------- = 2 bat. ----------- = 8 bat. 1 c. PB 1/2 c. sugar 3 eggs X 1 batch ------------- = 3 bat. 1 egg In this case, the limiting reagent is the PB since I only have enough to yield two batch of cookies. The sugar and the eggs are the ingredients that are in excess. Therefore, with the ingredients that I have at home, only 2 batches can be made. __________________________________________________________________________________________ |
Once you have your BCE (balanced chemical equation), there are several ways to figure out which substance is your limiting reagent.
The following BCE will be the equation used for the different methods that will presented:
C3H8 + 5O2 --> 3CO2 + 4 H2O (This is a combustion reaction and it is balanced)
10 g of propane gas ( C3H8) reacts with 10g of oxygen (O2)
Molar mass C3H8 : 3(12)g+8(1)g = 44g
Moral mass O2 : 2(16)g = 32 g
Molar mass CO2: 1(12)g+2(16)g = 44g
METHOD 1:
Use two separate equations and then compare the results obtained.
10 g C3H8 X 1 mol. C3H8 X 3 mol. CO2 X 44 g CO2
----------------- ---------------- -------------- = 30 g CO2 This means 30 g of CO2 can be produced if 10 g C3H8 is reacted
44 g C3H8 1 mol. C3H8 1 mol. CO2
10 g O2 X 1 mol. O2 X 3 mol. CO2 X 44 g CO2
-------------- ---------------- -------------- = 8.25 g CO2 This means only 8.25 g of CO2 can be produced if 10 g O2 is reacted.
32 g O2 5 mol. O2 1 mol. CO2 It also means that O2 is the limiting reagent!
METHOD 2:
Use one equation and compare the two reactants.
10 g C3H8 X 1 mol. C3H8 X 5 mol. O2 X 32 g O2
----------------- ---------------- -------------- = 36.4 g O2 This means if I reacted all 10 g of C3H8, I would need 36.4 g of O2.
44 g C3H8 1 mol. C3H8 1 mol. O2 Therefore, the limiting reagent is oxygen since I only have 10g of it.
The following BCE will be the equation used for the different methods that will presented:
C3H8 + 5O2 --> 3CO2 + 4 H2O (This is a combustion reaction and it is balanced)
10 g of propane gas ( C3H8) reacts with 10g of oxygen (O2)
Molar mass C3H8 : 3(12)g+8(1)g = 44g
Moral mass O2 : 2(16)g = 32 g
Molar mass CO2: 1(12)g+2(16)g = 44g
METHOD 1:
Use two separate equations and then compare the results obtained.
10 g C3H8 X 1 mol. C3H8 X 3 mol. CO2 X 44 g CO2
----------------- ---------------- -------------- = 30 g CO2 This means 30 g of CO2 can be produced if 10 g C3H8 is reacted
44 g C3H8 1 mol. C3H8 1 mol. CO2
10 g O2 X 1 mol. O2 X 3 mol. CO2 X 44 g CO2
-------------- ---------------- -------------- = 8.25 g CO2 This means only 8.25 g of CO2 can be produced if 10 g O2 is reacted.
32 g O2 5 mol. O2 1 mol. CO2 It also means that O2 is the limiting reagent!
METHOD 2:
Use one equation and compare the two reactants.
10 g C3H8 X 1 mol. C3H8 X 5 mol. O2 X 32 g O2
----------------- ---------------- -------------- = 36.4 g O2 This means if I reacted all 10 g of C3H8, I would need 36.4 g of O2.
44 g C3H8 1 mol. C3H8 1 mol. O2 Therefore, the limiting reagent is oxygen since I only have 10g of it.
|
METHOD 3: It's all about the mole ratio 10 g C3H8 X 1 mol. C3H8 ----------------- = 0.23 mol. C3H8 44 g C3H8 10 g O2 X 1 mol. O2 -------------- = 0.31 mol. O2 32 g O2 |
0.23 mol. C3H8 2.3 2 mol. C3H8 --------------------- = ----- Approx: ------------------ 0.31 mol. O2 3.1 3 mol. O2 The mole ratio presented here is : 2 mol. C3H8 : 3 mol. O2 This means 2 moles of C3H8 is required to react . with 3 moles of O2 (2/3=0.7) Whereas the true combining mole ratio is: 1 mol. C3H8 : 5 mol. O2 This means 1 mole of C3H8 is required to react with 5 moles of 02 (1/5=0.2) When comparing these mole ratios, we figure out that more O2 is required compared to what we actually have. Therefore it is the limiting reagent. Propane is in excess (XS). |
The three methods presented above all give the same answer. Use whichever method you prefer; they are all correct!
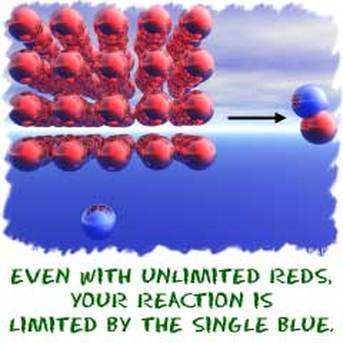
Now, look at the initial picture. Use the knowledge you have acquired to solve the problem. We see that we have 4 buns, 6 slices of cheese, 3 slices of lettuce, and 5 meat patties. In each sandwich we will need one of each. Our limiting reagent in this situation would be the lettuce since we have the least amount this ingredient. It is impossible to have 4 full hamburgers because on the fourth we would be missing a slice of lettuce.