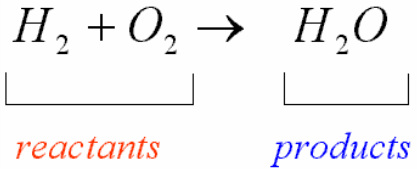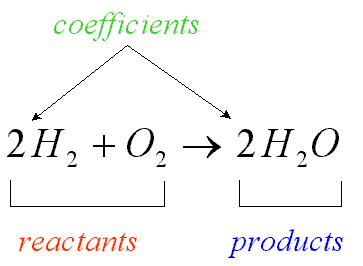When doing an experiment, you always know one thing is for sure: the mass of your reactants (left side of the chemical equation) will be equal to the mass of your products (right side of the chemical equation). This is known as the Law of Conservation of Mass which states that mass may never be created or destroyed, simply transferred or transformed.
Before balancing a chemical equation, you must first create one. Place the reactants on the left of your equation, the substances that are going to perform the chemical reaction. Then draw an arrow, which is like an equal sign but means "yield". Then finally, write the formulas for the products, the substances that were created during the chemical reaction. Place the correct subscripts next to the formulas and elements.
*Do not forget the seven diatomic elements: I2, H2, N2, Br2, O2, Cl2, and F2. These elements always have the subscript 2 because they are molecules composed of two atoms. An easy way to remember them is by saying this sentence: I Have No Bright Or Clever Friends.
Before balancing a chemical equation, you must first create one. Place the reactants on the left of your equation, the substances that are going to perform the chemical reaction. Then draw an arrow, which is like an equal sign but means "yield". Then finally, write the formulas for the products, the substances that were created during the chemical reaction. Place the correct subscripts next to the formulas and elements.
*Do not forget the seven diatomic elements: I2, H2, N2, Br2, O2, Cl2, and F2. These elements always have the subscript 2 because they are molecules composed of two atoms. An easy way to remember them is by saying this sentence: I Have No Bright Or Clever Friends.
Once we have this equation, we need to balance. Balance chemical equations is important because we need to assure ourselves that the Law of Conservation of Mass is obeyed. It also tells you the proportional quantities of each substance involved.
The coefficients can represent the number of molecules that react:
In this example we have 2 molecules of hydrogen reacting with one molecule of oxygen to produce 2 molecules of water.
We can also interpret these coefficients as the number of moles that react:
2 moles of hydrogen react with 1 mole of oxygen to produce 2 moles of water.
2 mol H2 X 2 g H2 1 mol O X 32 g O2
----------- + ------------ = 4g + 32 g = 36 g Reactants
1 mol H2 1 mol O2
2 mol H2O X 18 g H20
--------------- = 36 g Product The mass of reactants and the mass of products is the same. This satisfies
1 mol H20 the Law of Conservation of Mass.
In this example we have 2 molecules of hydrogen reacting with one molecule of oxygen to produce 2 molecules of water.
We can also interpret these coefficients as the number of moles that react:
2 moles of hydrogen react with 1 mole of oxygen to produce 2 moles of water.
2 mol H2 X 2 g H2 1 mol O X 32 g O2
----------- + ------------ = 4g + 32 g = 36 g Reactants
1 mol H2 1 mol O2
2 mol H2O X 18 g H20
--------------- = 36 g Product The mass of reactants and the mass of products is the same. This satisfies
1 mol H20 the Law of Conservation of Mass.
As a chemist student, you will not always be working with chemical equations as simple as the one provided above. Follow these steps when you encounter balancing these more difficult equations:
1) Balance all NON H's and O's
2) Balance all H's
3) Balance all O's
Example:
NaOH + H2SO4 --> NA2SO4 + H20
The four elements used in this equation are: Na (sodium), O (oxygen), H (hydrogen) and S (Sulfur).
1. Add up the number of atoms of each element has on the reactant side:
Na: 1 atoms O: 5 atoms H: 3 atoms S: 1 atoms
2. Add up the number of atoms each element has on the product side:
Na: 2 atoms O: 5 atoms H: 2 atoms S: 1 atom
3. Follow the rules from above. Start with balancing all the non H's or O's. In this case start with: Na, since S is already balanced
Stick a 2 coefficient in front of the Na on the reactants side because we see that there are two atoms of Na on the Product side.
The new equation looks like this :
2NaOH + H2SO4 --> NA2SO4 + H20
The Na's now match up
The 2 in front of the Na also affected the O and the H in that compound.
We now have in our reactants:
Na: 2 atoms O: 6 atoms H: 4 atoms ... and S remained unaffected: 1 atom
4. Balance the H's
Stick a 2 coefficient in front of the H20 compound on the product side.
The new equation looks like this :
2NaOH + H2SO4 --> NA2SO4 + 2H20
The H's match up.
We now have in our products:
Na: 2 atoms O: 6 atoms H: 4 atoms S: 1 atom
5. Double check everything is balanced:
Reactants:
Na: 2 atoms O: 6 atoms H: 4 atoms S: 1 atom
Products:
Na: 2 atoms O: 6 atoms H: 4 atoms S: 1 atom
THE EQUATION IS BALANCED!!!
____________________________________________________________________
To try more examples click on the button below:
1) Balance all NON H's and O's
2) Balance all H's
3) Balance all O's
Example:
NaOH + H2SO4 --> NA2SO4 + H20
The four elements used in this equation are: Na (sodium), O (oxygen), H (hydrogen) and S (Sulfur).
1. Add up the number of atoms of each element has on the reactant side:
Na: 1 atoms O: 5 atoms H: 3 atoms S: 1 atoms
2. Add up the number of atoms each element has on the product side:
Na: 2 atoms O: 5 atoms H: 2 atoms S: 1 atom
3. Follow the rules from above. Start with balancing all the non H's or O's. In this case start with: Na, since S is already balanced
Stick a 2 coefficient in front of the Na on the reactants side because we see that there are two atoms of Na on the Product side.
The new equation looks like this :
2NaOH + H2SO4 --> NA2SO4 + H20
The Na's now match up
The 2 in front of the Na also affected the O and the H in that compound.
We now have in our reactants:
Na: 2 atoms O: 6 atoms H: 4 atoms ... and S remained unaffected: 1 atom
4. Balance the H's
Stick a 2 coefficient in front of the H20 compound on the product side.
The new equation looks like this :
2NaOH + H2SO4 --> NA2SO4 + 2H20
The H's match up.
We now have in our products:
Na: 2 atoms O: 6 atoms H: 4 atoms S: 1 atom
5. Double check everything is balanced:
Reactants:
Na: 2 atoms O: 6 atoms H: 4 atoms S: 1 atom
Products:
Na: 2 atoms O: 6 atoms H: 4 atoms S: 1 atom
THE EQUATION IS BALANCED!!!
____________________________________________________________________
To try more examples click on the button below:
DO NOT FORGET TO ALWAYS MAKE SURE YOUR EQUATION IS BALANCED!