What is a Precipitate?
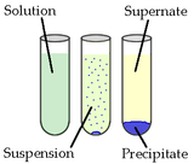
In Chemistry, a precipitate is a solid resulting from a chemical reaction. When you combine two or more different chemical substances the products are sometimes insoluble, unable to form a new solution, so it simply collects at the bottom of the beaker as a solid.
Why does this happen?
Like I just said, chemical reactions are magical things. You can start with two compounds and end up with two entirely new ones! Gas and heat and be produced, odours can occur, all this because of the different kinds of chemical reactions:
(where the letters represent different substances or solutions)
1) Synthesis, A + B --> AB
2) Decomposition, AB --> A + B
3) Single Displacement, AB + C = AC + B
4) Double Displacement, AB + CD = AD + BC
5) Combustion, Carbohydrate + O2 = CO2 + H20
So when all the reacting substances are switching partners in their chemical reaction, sometimes the products don't get along so well. Precipitates are most commonly formed after a double displacement reaction;
A (solution) + B (solution) --> C (solution) + D (solid) (precipitate)
Why does this happen?
Like I just said, chemical reactions are magical things. You can start with two compounds and end up with two entirely new ones! Gas and heat and be produced, odours can occur, all this because of the different kinds of chemical reactions:
(where the letters represent different substances or solutions)
1) Synthesis, A + B --> AB
2) Decomposition, AB --> A + B
3) Single Displacement, AB + C = AC + B
4) Double Displacement, AB + CD = AD + BC
5) Combustion, Carbohydrate + O2 = CO2 + H20
So when all the reacting substances are switching partners in their chemical reaction, sometimes the products don't get along so well. Precipitates are most commonly formed after a double displacement reaction;
A (solution) + B (solution) --> C (solution) + D (solid) (precipitate)
How does this happen?
How can we know whether the products will dissolve or not? There are two ways:
1) When writing your BCE's, refer to the RULES OF SOLUBILITY:
(these rules are to be applied in order)
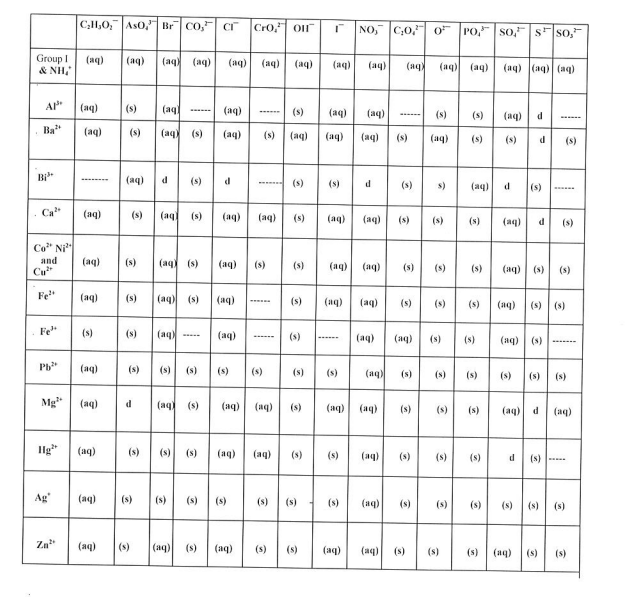
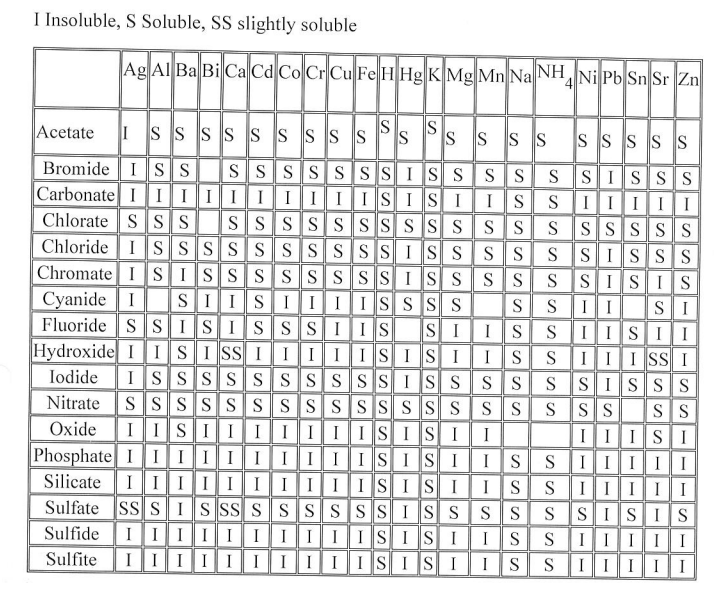
2) When writing your BCE's, refer to the SOLUBILITY CHARTS:
How can we know whether the products will dissolve or not? There are two ways:
1) When writing your BCE's, refer to the RULES OF SOLUBILITY:
(these rules are to be applied in order)
- All GR1A ions (alkali metals) and NH4 + (ammonium) compounds are soluble
- All NO3 - (nitrate), C2H402 + (acetate), ClO4 - (perchlorate), and ClO -3 (chlorate) ions are soluble
- All Lead, Mercury and Silver compounds are soluble
- Chlorides, bromides and iodides are all soluble
- All sulfates EXCEPT CALCIUM AND BARIUM are soluble
- Carbonates, Hydroxides, oxides, phosphates, silicates and sulfides are INsoluble
2) When writing your BCE's, refer to the SOLUBILITY CHARTS: